CV-AFS
Merx-T CV-AFS
The Merx-T Analyser from Brooks Rand uses the very sensitive technique of cold vapour atomic fluorescence spectrometry (CV-AFS) for the detection of total mercury in liquid samples. The low limit of detection (LOD) of 0.04 ng/L makes this instrument attractive for mercury monitoring in uncontaminated water samples. Mercury can also be determined in solid samples like sediments, plant and animal tissue when a suitable digestion or extraction technique is applied. User regulation
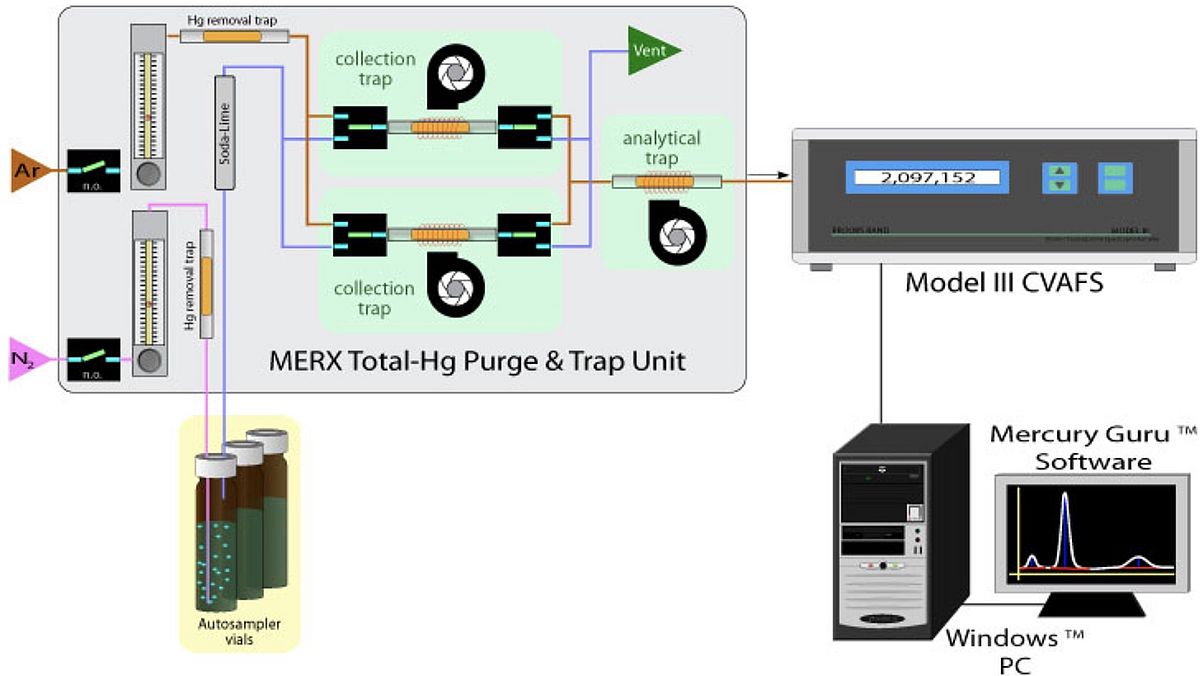

A water sample is acidified and stabilised with bromine monochloride in the field, a strong oxidation reagent which transforms both Hg(0) and organic mercury into Hg2+. In our laboratory, the excess of bromine monochloride is destroyed by the addition of hydroxylamine hydrochlorid and the subsequent addition of tin(II)chloride reduces Hg2+ to Hg(0). Hg(0) is a volatile atom which is driven out of the solution with nitrogen and collected on a gold trap. The Merx-T system works with a dual amalgamation step to remove interference compounds before they enter the detection chamber. By heating is the mercury released from the gold trap and enters the atomic fluorescence chamber. The outer shell electrons of the mercury atoms are excited with the UV light from a mercury-vapour lamp and emit the energy in form of fluorescence which is detected with a photomultiplier. The concentration of total mercury in the sample corresponds over a broad linear range with the atomic fluorescence signal.
