Isabel Lange, Marcos Toro, Rolf S.Arvidson, Inna Kurganskaya, Andreas Lüttge
Geochimica et Cosmochimica Acta (2021) 309, 329-351
https://doi.org/10.1016/j.gca.2021.06.032
Intrinsic heterogeneities of crystals almost always impose challenges on the investigation of dissolution kinetics. In the case of the incomplete solid solution series of alkali feldspars, additional complexity to the wide range in chemical composition and crystallographic characteristics is introduced in the form of chemical differentiation at low cooling temperatures. In this paper, we use direct measurements of intact crystal surfaces to quantify the dissolution rate of an alkali feldspar, providing details unavailable to powder dissolution experiments alone. In particular, we show how complex intergrowth textures characteristic of feldspars may play an important role in terms of how these minerals dissolve.
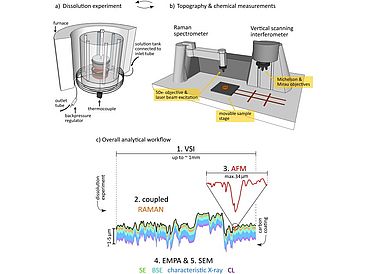
By means of a comprehensive experimental and analytical framework using Raman coupled vertical scanning interferometry (Rc-VSI), atomic force microscopy (AFM), light microscopy (LM), electron probe microanalysis (EPMA) and scanning electron microscopy (SEM), detailed information on heterogeneities in surface reactivity, morphology, chemistry, structure and texture of the dissolving surface are provided. The combination of spatially resolved qualitative and quantitative data revealed significant differences both in absolute rates and rate distributions associated with the potassium- and sodium-rich compositional compounds of which the latter dissolved slower with less dispersion in rates than the former. Whereas the spatial dissolution rate variances could not be explained by compositional differences between the two distinct feldspar phases alone, surface reactivity was successfully shown to vary with unique properties of the surface texture as well. A close relationship between enhanced dissolution and defect site distribution was observed, which critically depends on intergrowth geometry and phase internal twinning patterns. These observations are of great importance to any kind of experimental study and modeling approach which aims to improve the understanding of the dissolution kinetics of chemically and crystallographically complex materials.
© The Authors, https://doi.org/10.1016/j.gca.2021.06.032, licensed under CC BY-NC-ND 4.0