Electrochemical high-throughput characterization of metallic micro samples (SFB 1232)
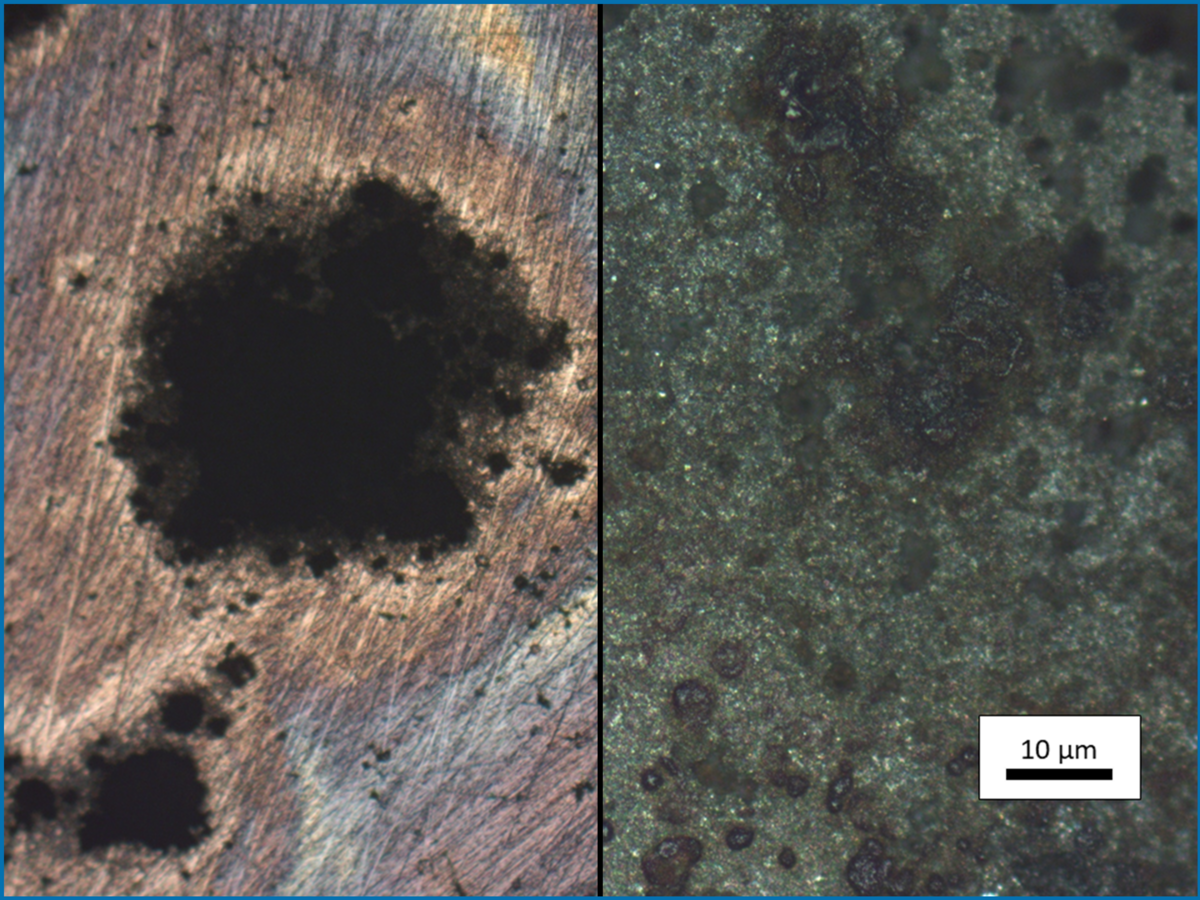
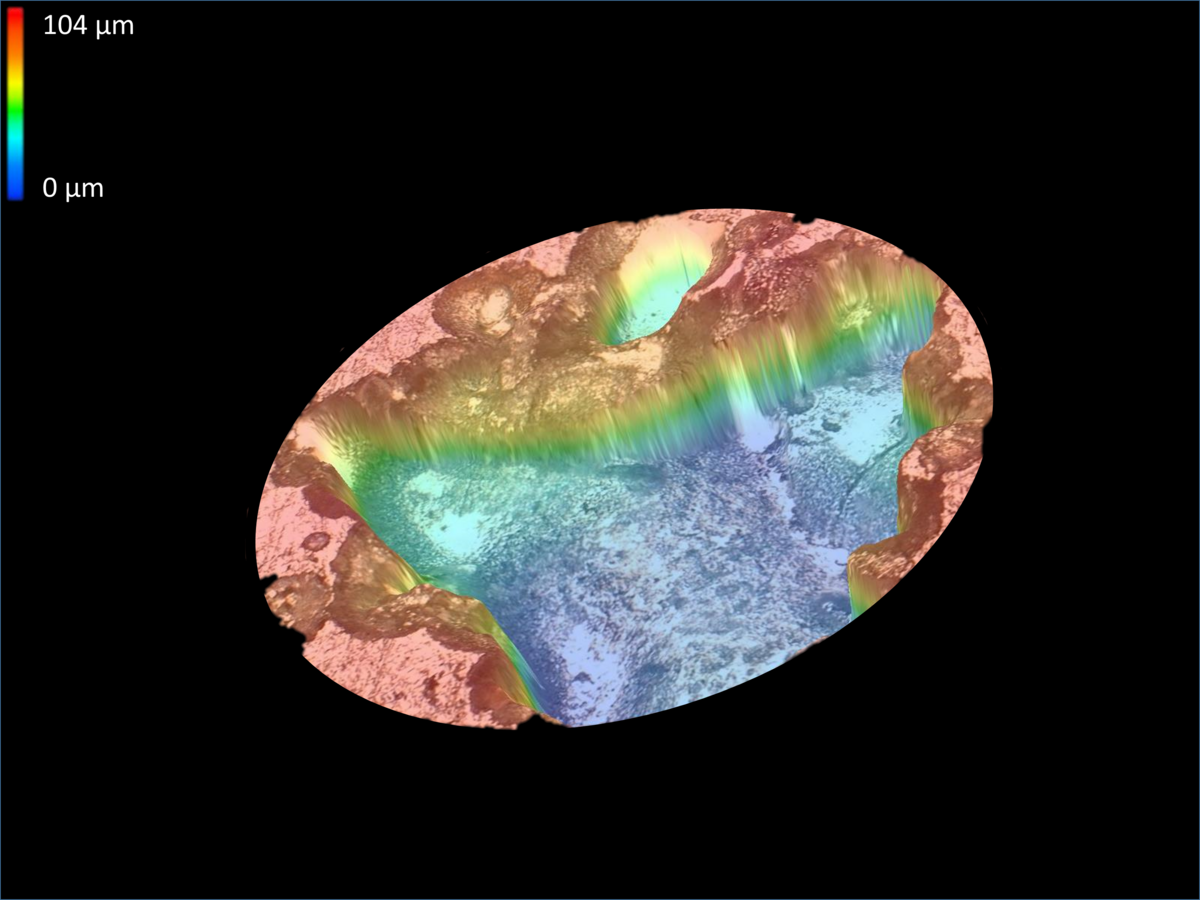
Metal corrosion refers to a process that involves degradation of the material functionality. The annual costs of corrosion damages amount to billions of euro. Corrosion phenomena, like passivity or pitting corrosion, can be determined by electrochemical characterization methods.
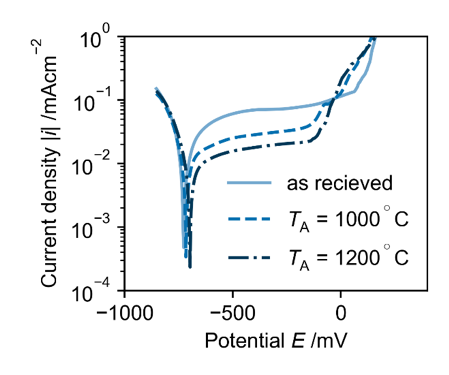
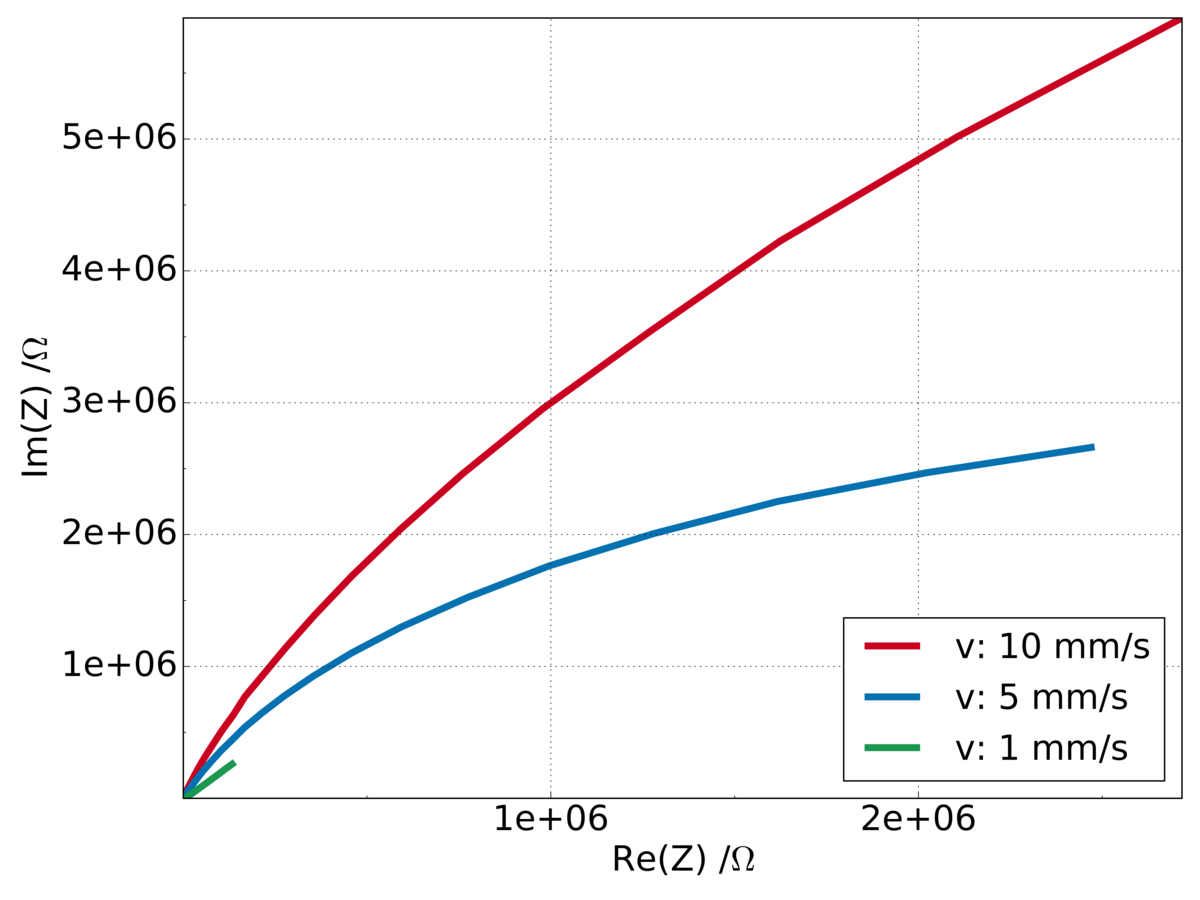
The aim of the project is the characterization of corrosion and material properties of micro samples by electrochemical methods like cyclic voltammetry, impedance spectroscopy and electrochemical noise technique. One main research question is the correlation of electrochemical measurements on micro samples in short time periods (seconds or minutes) and corrosion phenomena on metallic components in real time (several years). An additional research focus are the effects of changes in the micro structure on the ability to form protective passive layers.
The project ‘Electrochemical high-throughput characterization of metallic micro samples’ is part of the Collaborative Research Center SFB 1232 ‘Farbige Zustände’ about the development of new high-performance materials. As part of the Collaborative Research Center micro samples of different composition and treatment have to be generated and characterized. This project is responsible for the electrochemical characterization of the samples and passivity is of particular interest.
The passivity of iron-based materials is caused by a formation of an oxide layer on the surface of the metal. This passive layers can protect the metal surface against corrosion attacks. The stability of the passive layer is related to the composition and the micro structure of the alloy. A possibility to improve the passive layers through micro structure optimization has to be explored by high throughput electrochemical measurements.
Relevant publications
Ingmar Bösing's PhD thesis
Bösing, I. et al. (2020) Corros. Mater. Degrad. 1(1), 6. https://doi.org/10.3390/cmd1010006
S. Imani Moqadam et al. (2020) Metals, 10(3), 297. https://doi.org/10.3390/met10030297
I. Bösing et al. (2020) Int. J. Electrochem. Sci. 15, 319 – 333. http://doi.org/10.20964/2020.01.09
M. Steinbacher et al. (2019) High-Throughput 8(4), 22. https://doi.org/10.3390/ht8040022
I. Bösing et al. (2019) AIP Advances 9, 065317. https://doi.org/10.1063/1.5094615
I. Bösing et al. (2018) MATEC Web of Conferences 190, 04002, http://doi.org/10.1051/matecconf/201819004002.
I. Bösing et al. (2017) International Journal of Corrosion 9425864, http://doi.org/10.1155/2017/942586.
Relevant files
Contact:
Dr.-Ing. Ingmar Bösing
Room UFT 2220
Fon 0421 - 218 - 63466